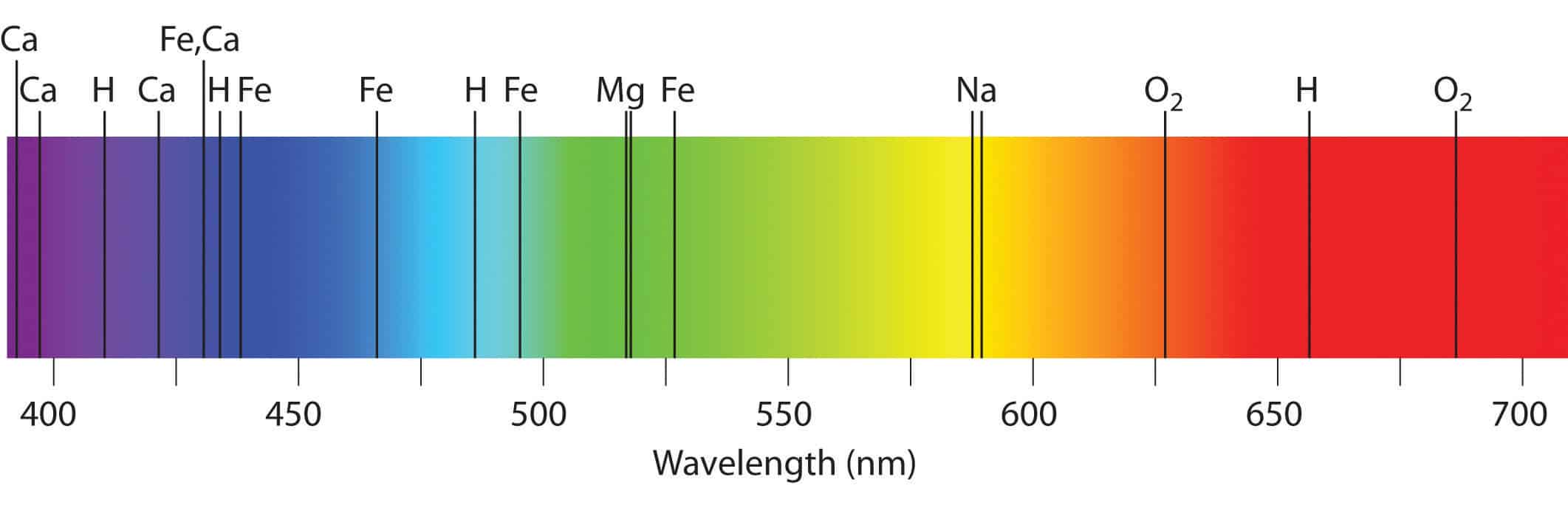
The result of Atomic Emission Spectroscopy is an emission spectrum which is specific. For energies $E 0$, you have a continuum of scattering states. This pattern of colours is known as a continuous spectrum. 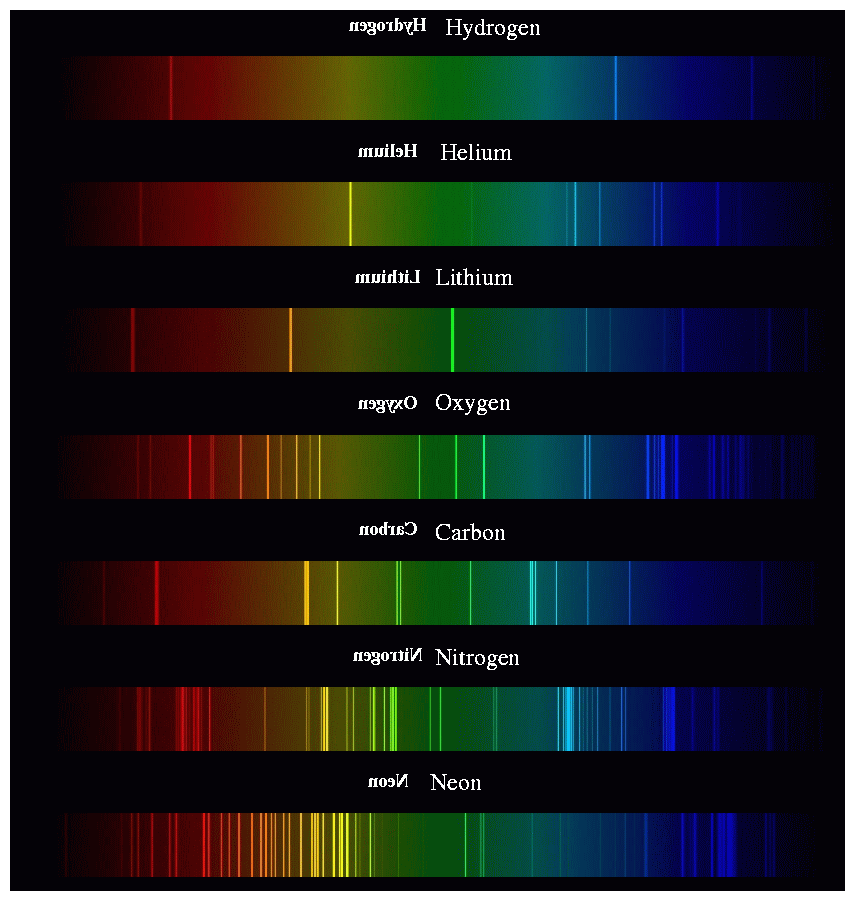
Acting on any real exponential with the second derivative just gives you the same function multiplied by a positive factor, so the overal sign is negative. However, if one confines the wavefunction in the segment $$ one has that $\psi(0)=\psi(L)$ which gives $e^ + V(x) )\psi = E \psi$ and note that in the region where $V(x) \rightarrow 0$, you can disregard the second term in the Hamiltonian. The wavefunction has a continuous of energy levels $\omega\propto k^2$. 
The spectrum of light that a cloud of gas emits depends on its temperature, density. Emission Spectrum: Starlight can also heat up a cloud of gas, exciting the atoms and molecules within the gas, and causing it to emit light. Which describes the wavefunction of a free particle (or the propagation of a sinusoidal wave of a classical field). An absorption spectrum has dark lines or gaps in the spectrum corresponding to wavelengths that are absorbed by the gas. Therefore in the thermodynamical limit (large system sizes), these quantized energy levels become a continuum of states, since $\Delta\rightarrow0$.Īs an example, let us consider a plane wave In a solid, energy levels are also quantized, but the difference $\Delta$ between levels decreases as the system size increases. The light - in this case, from an ordinary light bulb - has been spread out into different colors. This lab explores some of the basic ideas used to analyze spectra. In the quantum realm, energy levels are quantized if the wavefunction is confined in a finite space, e.g., in an atomic orbital or in a quantum well. An absorption spectrum occurs when light passes through a cold, dilute gas and atoms in the gas absorb at characteristic frequencies since the re-emitted light. The key is to spread the light out by color, producing a spectrum like the one shown in Fig. In such a string, the allowed "energy states" corresponds to frequencies (harmonics) which are multiples of a fundamental frequency (first harmonic). The electromagnetic spectrum is a continuous distribution of wavelengths ranging from ultraviolet to infrared radiation. For example, consider the allowed frequencies of a string with fixed length (confinement), as in a guitar or violin. On the other hand, also classical systems exhibits quantization of the energy levels. In fact, anytime that a wave equation (any quantum equation for the wavefunction, or a classical equation for a classical field, e.g., EM field) has periodic boundary conditions in some spatial variables, the system exhibits quantized energy levels.Īs noticed in the question and in other answers, energy levels in quantum systems are not always quantized. Q-2: The atomic spectrum of Li 2+ should be similar to that of. Hence, the line spectrum is also called atomic spectrum.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
